Seed germination is the prelude to the blooming of life. The oil stored in the cotyledons or endosperm serves as the core energy source driving this miraculous life process. During germination, oils undergo a series of complex metabolic transformations, with their content and distribution changing dynamically. To accurately capture these changes, selecting an appropriate analytical method—namely, an oil analysis method—is crucial.

A study published in the Journal of the Science of Food and Agriculture by a team from Northwest A&F University systematically compared the advantages and disadvantages of three oil detection methods using peanut, soybean (herbaceous oilseeds), camellia seed, and almond (woody oilseeds) as test subjects, and validated the reliability and green attributes of low-field nuclear magnetic resonance (LF-NMR) technology. Today, based on this study, we explain the “skill points” and “applicable scenarios” of different oil detection methods.
01 Why is the traditional Soxhlet extraction called the “Gold Standard”, yet not “universal”?
The status of the Soxhlet extraction method stems from its solid principle and accurate measurement.
An organic solvent (e.g., petroleum ether) is used in a reflux apparatus to continuously extract oil from the sample. The oil content is calculated by weighing the sample before and after extraction.
This is a classical method for determining total oil content, providing direct and accurate results, and serving as a benchmark for validating the reliability of other new methods.
Destructive: The sample must be pulverized, making it impossible to continuously observe the same seed.
Time‑consuming: A single extraction typically takes several hours (10 hours in this study).
Organic solvent consumption: Requires large amounts of organic reagents, resulting in poor environmental friendliness.
Single information: Only total oil content is obtained; spatial distribution information cannot be acquired.
When your core goal is to obtain the precise total oil content of a sample and you do not mind destroying the sample, this is the preferred choice—for example, routine quality testing or calibration for modeling other methods.
02 What microscopic world does Confocal Laser Scanning Microscopy (CLSM) open up for us?
CLSM takes our vision to the cellular level, allowing us to directly “see” oil bodies.
Lipids are labeled with a specific fluorescent dye (e.g., Nile Red). Under excitation by a laser of a specific wavelength, oil bodies emit red fluorescence, enabling high‑resolution, high‑contrast imaging of oil bodies within individual cells.
Ultra‑high resolution: The morphology, size, and distribution of oil bodies can be clearly observed – a capability unmatched by other methods.
Strong specificity: Directly targets lipids through staining, providing a clear signal.
Destructive sample preparation: Requires cryo‑sectioning, fixation, and staining of seed tissues. The procedure is complex and does not allow dynamic monitoring of living samples.
Phototoxicity and photobleaching: Prolonged excitation can affect sample activity or cause fluorescence signal decay.
High cost and technical barrier: Instruments are expensive to purchase and maintain, requiring specialized skills.
Complex quantification: Although image analysis software can provide oil area ratios, differences in sample preparation easily introduce errors. Environmental friendliness is also low.
When your research needs to investigate the fine distribution of oil within tissue cells, changes in oil body size, or interactions with other organelles, CLSM is an indispensable and powerful tool. It answers the micro‑mechanistic question: “Where is the oil and what is its morphology?”

Figure 1. Changes in oil distribution during germination. (b) CLSM images. Peanut and camellia were selected as representatives of herbaceous and woody seeds, respectively.
03 How does Low‑Field Nuclear Magnetic Resonance (LF‑NMR) enable non‑destructive, continuous monitoring?
LF‑NMR acts like a non‑invasive “scanner” by sensing hydrogen proton signals, allowing rapid detection of intact seeds.
Hydrogen protons in the sample (mainly from water and oil) undergo relaxation after being excited in a magnetic field. Hydrogen protons in different physicochemical environments have different transverse relaxation times (T₂). By analyzing the T₂ relaxation spectrum, signals from bound water, free water, and oil can be distinguished and quantified.
Truly non‑destructive: No pretreatment is required; intact, viable seeds can be directly measured – the foundation for dynamic continuous monitoring.
Fast and efficient: A single scan takes only a few minutes.
Rich information dimensions: The T₂ peak area quantitatively reflects oil content; the T₂ relaxation time reflects oil fluidity (related to fatty acid unsaturation and carbon chain length).
Green: No organic solvents are used, making it the greenest among the three methods.
It cannot provide cellular‑level resolution like CLSM, nor can it directly give absolute mass percentages (a calibration model is required).
When your research focuses on “dynamic change processes”, such as continuously monitoring oil changes in the same batch of seeds during germination, processing, or storage over several days, LF‑NMR is currently the only ideal choice. It answers the process question: “How does total oil content and oil state change over time?”
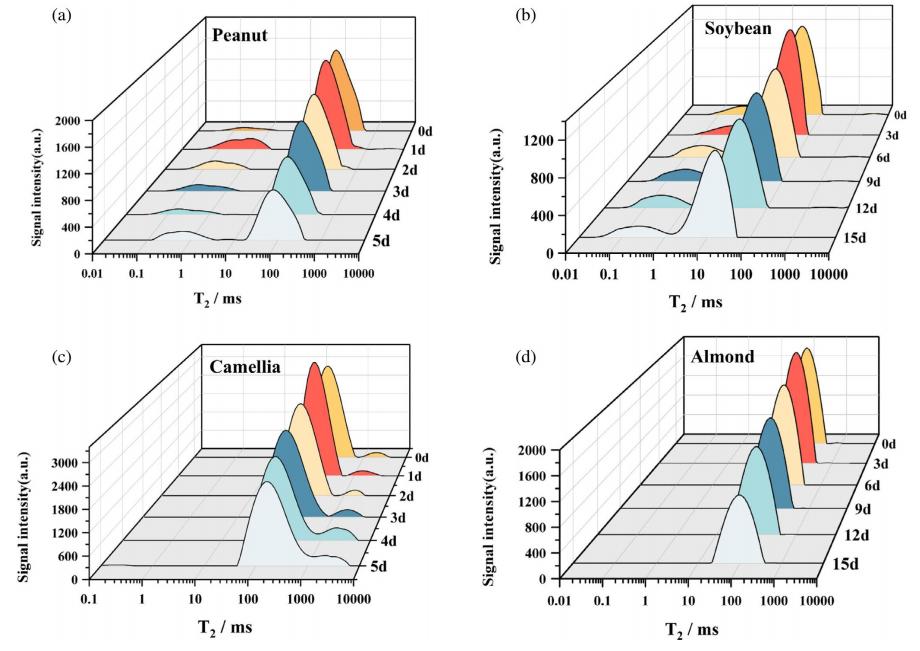
Figure 2. Changes in transverse relaxation time (T₂) distribution of different seeds during germination. (a) Peanut; (b) Soybean; (c) Camellia; (d) Almond.
04 How does Magnetic Resonance Imaging (MRI) allow us to intuitively “see” the oil map inside seeds?
MRI spatially encodes the aforementioned proton signals to generate a pseudo‑color image resembling a “heat map”, where blue/green represent low signals and red/yellow represent high signals, intuitively displaying the macroscopic distribution of oil within the seed.
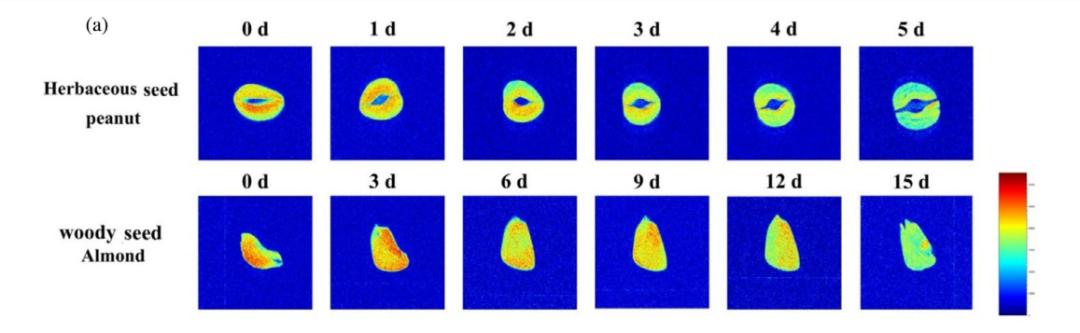
Figure 3. Changes in oil distribution during germination. (a) MRI images; peanut and camellia were selected as representatives of herbaceous and woody seeds, respectively.
The advantage lies in perfectly combining oil content information with spatial distribution information. The Northwest A&F University team’s results showed that the oil area ratio from MRI images exhibited the highest correlation with actual oil content (R² > 0.9), and the accuracy of its predictive model was even better than models built from CLSM images and T₂ spectra.
It answers the question: “In which specific part of the seed (e.g., cotyledon, hypocotyl) is oil being consumed or accumulated?” Combined with LF‑NMR relaxation spectra, the two constitute a complete analytical solution from “macroscopic distribution” to “physical state”.
05 Let the research method serve the scientific question
Choosing which method depends on the scientific question we want to answer:
Want to know “Total oil content”? → Soxhlet extraction is the first choice.
Want to explore “What do oil droplets look like in cells and where are they distributed?” → Choose CLSM.
Want to track “How does oil in the same seed change over its life cycle?” → LF‑NMR (T₂ relaxation analysis) is the only solution.
Want to visually see “The map of oil growth and depletion in different tissues inside the seed?” → LF‑NMR combined with MRI is an irreplaceable non‑invasive approach.
The choice of method always serves the scientific question. Traditional Soxhlet extraction and CLSM remain the “gold standards” for specific dimensions. However, the emergence of LF‑NMR/MRI technology is not a simple replacement of older methods, but an expansion of the research paradigm – it empowers us to ask questions that could not be dynamically observed in the past. The study also used AGREEprep and MoGAPI tools to evaluate the greenness of the three methods, and the results consistently showed that LF‑NMR, because it completely abandons organic solvents, requires no complex pretreatment, and is non‑destructive and rapid, is the most environmentally friendly solution.
Reference
《Optimization of LF-NMR-based methods for analysis of oil content and distribution in germinating oilseeds》
Phone: 400-060-3233
After-sales: 400-060-3233


Back to Top