Background
Agarose is a natural linear polysaccharide derived from red algae, composed of alternating β‑1,3‑galactose and α‑1,4‑3,6‑anhydro‑L‑galactose units. It dissolves in hot water and forms a thermally reversible three‑dimensional hydrogel upon cooling. Agarose has been widely used in the food industry, bioseparation, cell culture, tissue engineering, and 3D bioprinting.
As a typical physically crosslinked natural polysaccharide gel, agarose aqueous solution undergoes a sol‑gel transition upon cooling and reverts to the solution state upon heating, exhibiting a pronounced thermal hysteresis exceeding 30 °C. This unique property makes agarose highly promising for smart sensing and memory devices. Despite decades of research, the molecular mechanisms underlying its gelation and thermal hysteresis remain incompletely understood.
To address this challenge, Professor Peiyi Wu’s group at Donghua University systematically investigated the molecular mechanism of the thermally reversible sol‑gel transition of agarose in water from the perspective of water dynamics, using a low‑field nuclear magnetic resonance (LF‑NMR) instrument (model VTMR20‑010V‑I) manufactured by Suzhou Niumag Analytical Instrument Corporation.
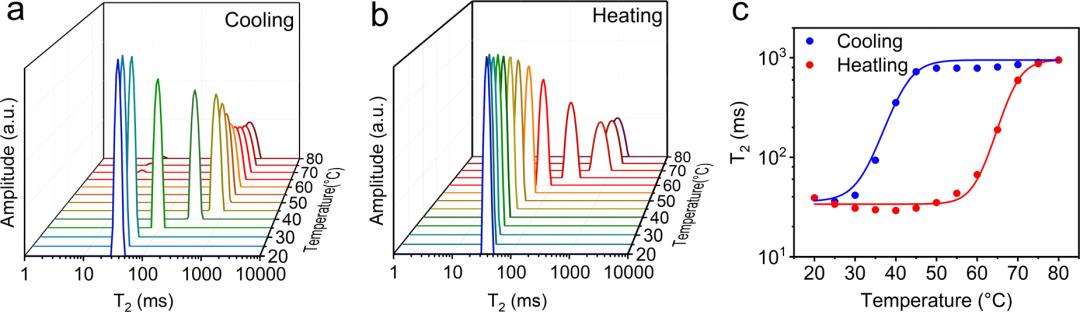
Figure 1: (a) Variable‑temperature T₂ relaxation spectra of agarose aqueous solution during cooling (sol → gel) from 20 °C to 80 °C; (b) during heating (gel → sol); (c) Transverse relaxation time (T₂) as a function of temperature during cooling and heating, directly demonstrating thermal hysteresis.
Figure 1a – Cooling process (sol → gel)
At high temperature (~80 °C): the T₂ relaxation time is very long (>100 ms), indicating that water molecules are highly mobile and the system is in the sol state.
When the temperature drops to ~35 °C: T₂ decreases sharply, water molecules rapidly change from a free state to a restricted state, marking the onset of the sol‑gel transition.
At low temperature (20 °C): T₂ remains at a very short level, water molecules are tightly bound by the three‑dimensional agarose network, and the system is fully gelled.
Figure 1b – Heating process (gel → sol)
At low temperature (20 °C): T₂ remains short, water molecules are still restricted, and the gel structure is stable.
Before the temperature reaches 65 °C: T₂ remains almost unchanged, indicating no significant disruption of the gel network.
When the temperature exceeds 65 °C: T₂ suddenly rises sharply, water molecules regain free motion, and the system undergoes a gel‑sol transition.
Figure 1c – T₂ as a function of temperature (most critical, direct proof of hysteresis)
Cooling curve: transition point at ~30–35 °C.
Heating curve: transition point at ~65 °C.
The two curves do not overlap, forming a clear hysteresis loop.
This directly demonstrates that agarose hydrogels exhibit a significant thermal hysteresis effect (~40 °C). Once the gel is formed, the hydrogen bonds between agarose chains are highly stable and require a high temperature to break; therefore, water molecules do not regain freedom until high temperatures are reached.
Reference
Zhu W Y, Hou L, Wu P Y. On the molecular mechanism of thermally reversible sol‑gel transition with pronounced hysteresis of agarose in water[J]. Soft Matter, 2025, 21: 7091‑7099. DOI: 10.1039/D5SM00630A.
Phone: 400-060-3233
After-sales: 400-060-3233


Back to Top