Surimi products, represented by fish balls, are prone to protein denaturation, texture deterioration, and juice loss during frozen storage due to ice crystal formation and recrystallization. The damage to myofibrillar structure and water distribution caused by ice crystals is the core reason for quality degradation and a key bottleneck restricting the industrial upgrading of traditional fish balls. Conventional cryoprotectant systems often rely on phosphates and sugars. However, against the backdrop of “clean label” and health demands, there is an urgent need to develop novel cryoprotectants from natural sources with minimal sensory impact.
Short-chain polypeptides (SCPs), characterized by their low molecular weight, abundant polar groups, and properties such as adsorption-inhibition activity and thermal hysteresis effect, show application potential as natural cryoprotectants in the frozen food sector. Nevertheless, the mechanism by which SCPs regulate ice crystal growth in fish ball systems remains unclear, particularly their specific influence on the state and migration behavior of water within the fish ball gel network. Direct evidence at the molecular level is still lacking.
This study prepared SCPs by enzymatically hydrolyzing bovine hide gelatin using compound protease. The molecular weight of the obtained SCPs was predominantly distributed between 180-3000 Da (accounting for 86.16%). This specific molecular weight range endows them with excellent molecular flexibility and hydrophilicity.
This study utilized LF-NMR technology to clearly reveal the impact of SCPs on the state and migration behavior of water in fish balls during frozen storage from two dimensions: water state distribution and spatial distribution. The core value of LF-NMR lies in its ability to precisely quantify water phase transitions, visualize water distribution, and corroborate these findings with physicochemical indicators, thereby providing direct molecular evidence for the cryoprotective effect of SCPs.
The research employed the CPMG pulse sequence to acquire transverse relaxation time (T2) data. Inversion algorithms were then applied to analyze the signals, obtaining dynamic evolution information of different water fractions during the freezing process. Furthermore, MRI imaging results, along with their threshold-segmented images, visualized the spatial distribution of water within the fish balls.
Figure 1 presents the core results obtained through LF-NMR analysis in this study. It intuitively compares the dynamic changes in water state and distribution in fish balls with different SCP additions before and after frozen storage, using T2 relaxation time distribution spectra and Magnetic Resonance Imaging (MRI).
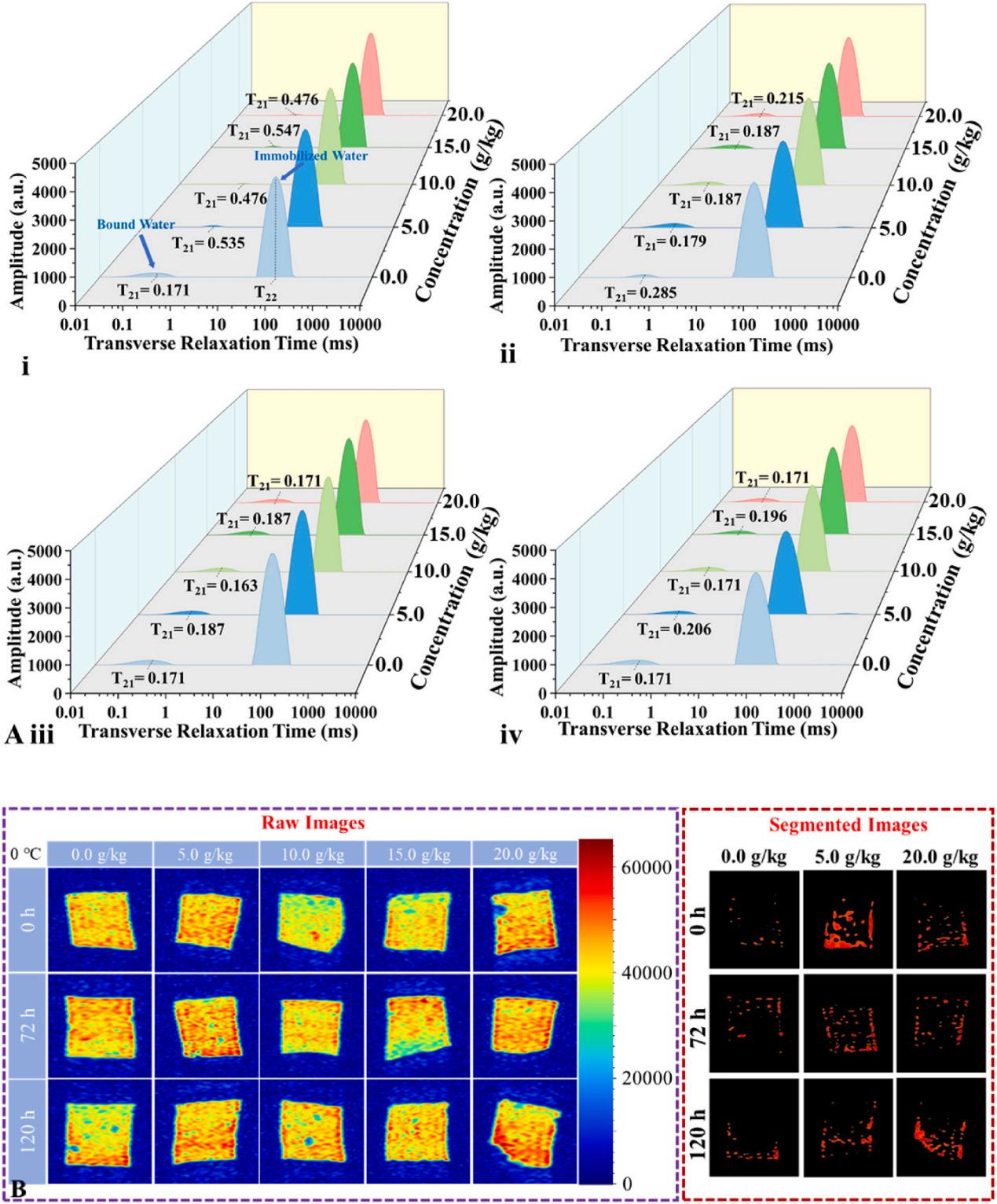
Figure 1. Dynamic changes in water state and distribution in fish balls with different SCP additions before and after frozen storage: (A) T2 relaxation spectra of fish balls with varying SCP additions; (B) MRI images and their corresponding threshold-segmented images of fish balls after different storage times at -20°C (red signal areas represent high moisture content).
Part.1 T2 Relaxation Spectrum: Revealing Water State Transition Patterns
By analyzing the T2 relaxation time (x-axis, unit: ms) and signal amplitude (y-axis), LF-NMR clearly delineated the water in fish balls into two main peaks:
Key Findings:
Pre-freezing Competitive Binding: Upon addition of SCPs, the T21 relaxation time shifted to the right, and its corresponding peak area proportion (A21) decreased. This indicates that SCPs participate in reshaping water-protein interactions. The polar groups of SCPs competitively bind to hydrophilic sites on myofibrillar proteins, inducing a migration of bound water (T21) towards immobilized water (T22). This enhances the mobility of bound water and optimizes the initial water distribution.
Post-freezing Mitigation of Water Loss: After frozen storage, the control group without SCPs showed a decrease in A22 (the area proportion of the immobilized water peak), indicating gel network disruption and water loss caused by ice crystal growth. In contrast, SCP-treated groups better maintained A22. From the perspective of water state dynamics, this demonstrates the ability of SCPs to inhibit ice crystal damage and protect the gel network.
Furthermore, LF-NMR data confirmed that 10.0 g/kg of SCPs could shorten the T21 relaxation time from 0.285 ms in the control group to 0.187 ms, significantly reducing the water migration rate during frozen storage.
Part.2 MRI Imaging: Visualizing Spatial Water Distribution
In the MRI results, red signal regions denote high moisture content, while blue signal regions indicate low moisture content.
Part.3 Mechanistic Insights: A Model of Water Regulation Based on LF-NMR
Integrating LF-NMR, SEM, and physicochemical indices, this study proposes a dual mechanism:
01 Competitive Binding Mechanism
SCPs, rich in carboxyl, amino, and hydrophobic groups, can interact with the system. At an optimal addition level (10.0 g/kg), they optimize water distribution. However, excessive addition (≥15.0 g/kg) can interfere with hydrophobic interactions and disulfide bond formation between proteins, leading to a significant decline in rheological parameters (storage modulus G′, loss modulus G′′). This manifests as a looser gel network and increased cooking loss (reaching 13.1% in the 20.0 g/kg group).
02 Adsorption-Inhibition Mechanism
SCPs adsorb onto the surface of ice crystals via hydrogen bonds, inhibiting their growth and inducing a Thermal Hysteresis (TH) effect. This results in a ‘gap’ in ice crystal growth and the formation of characteristic pyramidal ice crystal structures. This microstructural modification reduces the mechanical disruption caused by ice crystals to the myofibrillar protein network, thereby maintaining structural integrity after frozen storage.
This study, leveraging LF-NMR technology, elucidated the dual mechanism of short-chain polypeptides during the frozen storage of fish balls at the molecular level: firstly, altering the initial water state through competitive binding, and secondly, protecting the gel network by inhibiting ice crystal growth, thereby reducing the loss of immobilized water and maintaining product quality.
Experiments demonstrated that 10.0 g/kg was the optimal addition level. At this concentration, after 120 hours of frozen storage at -20 °C, the fish balls exhibited a 10.8% reduction in cooking loss compared to the additive-free group, an 11.8% improvement in color stability (b* value), and received sensory evaluations closest to fresh samples.
In practical applications, LF-NMR technology provides data support for the precise optimization of frozen food formulations (such as determining the optimal addition level of SCPs), improvement of freezing processes, and product quality control. It offers a reliable technical approach for studying the phase behavior of water and quality regulation in other complex food systems.
Reference:
Xu, Y., Guo, X., Zhao, L., Ma, H., Zhang, L., & Fan, F. (2026). Short-chain polypeptides modulate ice crystallization to enhance cryoprotection of Saurida tumbil-derived fish balls. LWT, 119031. https://doi.org/10.1016/j.lwt.2026.119031
Research Team
Associate Professor Fanghui Fan’s Team, Shenzhen University
Research Focus
“Regulation of Frozen Fish Ball Quality by Short-Chain Polypeptides Analyzed via LF-NMR Technology”
Journal
LWT
Impact Factor
JCR Category (Chinese Academy of Sciences): Agricultural and Biological Sciences (Category 1)
Phone: 400-060-3233
After-sales: 400-060-3233


Back to Top