Background
Poly(ethylene-co-methacrylic acid) ionomers exhibit ionic aggregation structures and chain segment mobility during melt processing, which directly influence flowability, processability, and final product performance. However, traditional characterization methods face the following challenges in molten-state, high-viscosity systems:
Inability for in-situ characterization: Methods like SAXS and TEM require solid samples and cannot reflect the dynamic structure in the melt state.
Long testing cycles: Rheological tests can reflect macroscopic viscosity but cannot resolve microscopic phase distribution.
Insensitivity to high-viscosity samples: EMAA melts with a high degree of neutralization (DoN > 60%) exhibit excessively high viscosity, making measurement difficult with traditional rheometers.
Inability to distinguish dynamic phases: Methods such as DSC and FTIR struggle to differentiate the mobility differences among “ion-rich aggregates”, “intermediate phases (tie chains)”, and “high-mobility phases (dangling chains)”.
Solution: A Low-Field NMR Strategy for Rapid, In-situ, Multi-Phase Resolved Characterization
Currently, in a paper published by Dow Chemical, a low-field time-domain NMR (TD‑NMR) instrument (Model VTMR20-010V-I) from Niumag Analytical Instrument Corporation, combined with the CPMG pulse sequence and a three-component fitting model, was used for rapid, non-destructive in-situ analysis of EMAA above its melting temperature (100°C). A single test simultaneously provides:
✅ Proportion of three dynamic phases (Aˡ, Aⁱ, Aʰ)
✅ Mobility indicators for each phase (T₂ˡ, T₂ⁱ, T₂ʰ)
✅ Comprehensive indicator for overall chain mobility (T₂ᵃᵛᵍ)
EMAA is classified into a low-mobility phase (ion aggregates, acid groups and their surrounding polymer chains; superscript l), an intermediate phase (flexible network chains between ion clusters and acid groups; superscript i), and a high-mobility phase (dangling chains, low molecular weight oligomers; superscript h).
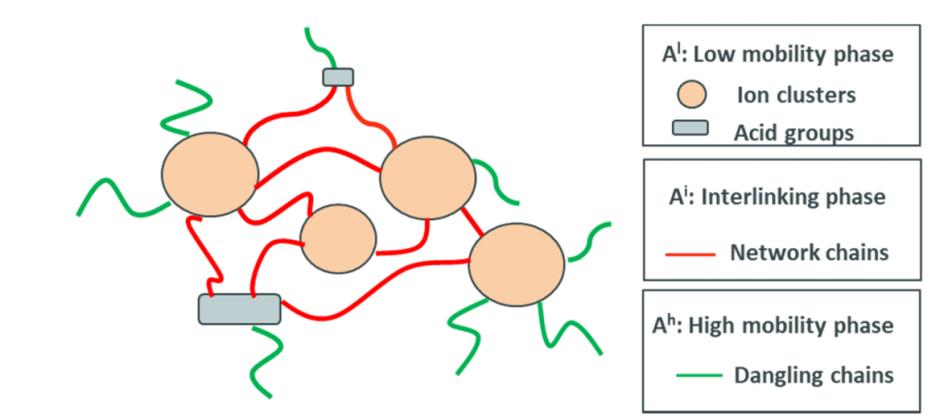
Figure 1: Schematic diagram of the dynamic phase composition model for EMAA ionomers
Part 1: Effect of Degree of Neutralization (DoN) on Dynamic Phases and Chain Mobility
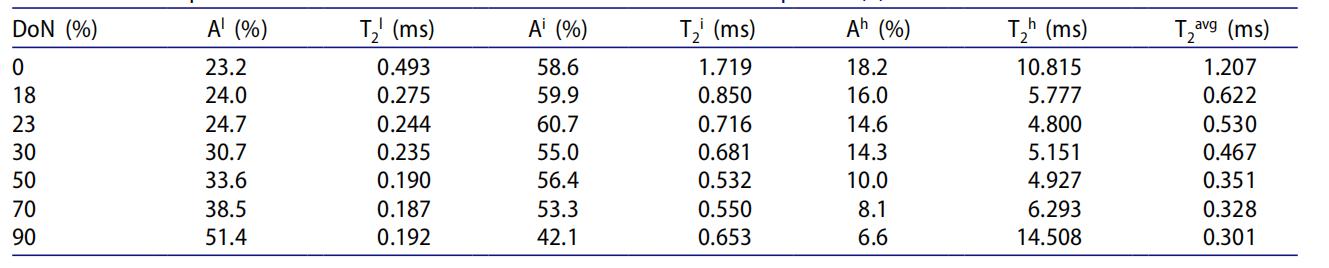
Table 1: Effect of Degree of Neutralization (DoN) on Dynamic Phases and Chain Mobility
Low-Mobility Phase (Aˡ, T₂ˡ):
Aˡ continuously increases with rising DoN (23.2% at 0% DoN → 51.4% at 90% DoN), indicating that higher neutralization leads to more ion aggregate formation. T₂ˡ decreases initially with increasing DoN and then stabilizes (0.493 ms at 0% DoN → 0.190 ms at 50% DoN), remaining essentially constant at 0.187-0.192 ms beyond 50% DoN, suggesting the rigidity of ion aggregates increases with DoN and reaches a packing limit around 50% DoN.
Intermediate Phase (Aⁱ, T₂ⁱ):
Aⁱ shows an initial slight increase (0%-23% DoN) followed by a decreasing trend, with inflection points at 23% and 50% DoN, reflecting the balance between ion aggregate formation and network chain transformation. T₂ⁱ decreases initially with increasing DoN (1.719 ms at 0% DoN → 0.532 ms at 50% DoN) and then shows a slight increase (0.532→0.653 ms from 50% to 90% DoN). This indicates that at low DoN, network chain mobility is restricted by entanglement with ion aggregates, while at high DoN, the capture of some low-mobility short chains by ion aggregates leaves remaining longer chains with slightly enhanced mobility.
High-Mobility Phase (Aʰ, T₂ʰ):
Aʰ continuously decreases with increasing DoN (18.2% at 0% DoN → 6.6% at 90% DoN), showing that more dangling chains are incorporated into the low-mobility phase. T₂ʰ decreases initially with increasing DoN (10.815 ms at 0% DoN → 4.927 ms at 50% DoN) and then increases significantly (14.508 ms at 90% DoN). Beyond 50% DoN, phase separation between the high-mobility and low-mobility phases intensifies, reducing restrictions on dangling chains.
Overall Mobility (T₂ᵃᵛᵍ):
Continuously decreases with increasing DoN (1.207 ms at 0% DoN → 0.301 ms at 90% DoN), consistent with the known characteristic of increasing EMAA melt extensional viscosity with higher DoN, validating the reliability of the low-field NMR data.
Technical Value: A single TD‑NMR test systematically reveals the impact of DoN on the evolution of the three-phase structure, providing direct guidance for balancing “degree of neutralization – processability” in formulations.
Part 2: Effect of Plasticizers on Dynamic Phase Distribution and Chain Mobility
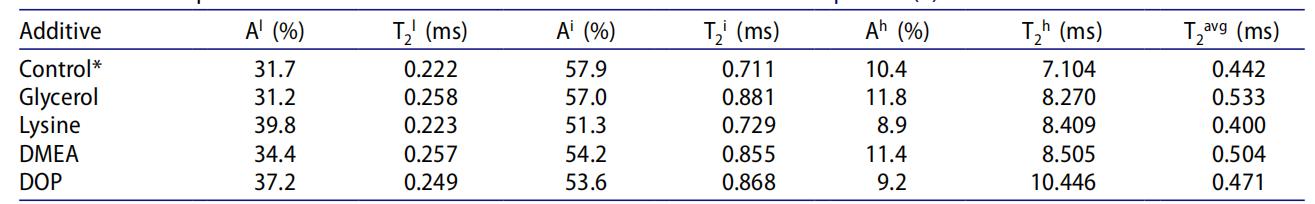
Table 2: Effect of Plasticizers on Dynamic Phase Distribution and Phase Mobility (Control group is a new batch of EMAA sample with 27.5% DoN.)
This table investigates the effects of four plasticizers on the dynamic phases and chain mobility of EMAA. The results are analyzed as follows:
Control Group:
Serves as the benchmark with T₂ᵃᵛᵍ = 0.442 ms. The phase content and T₂ values provide a baseline for comparison with plasticizer effects.
Glycerol:
Aˡ remains essentially unchanged (31.7% → 31.2%), T₂ˡ increases (0.222 → 0.258 ms), T₂ᵃᵛᵍ increases significantly (0.442 → 0.533 ms). Due to its polarity and hydrogen bonding, glycerol inserts into ion aggregates, loosening their packing structure, reducing rigidity, and enhancing overall chain mobility without forming new low-mobility phases.
Lysine:
Aˡ increases significantly (31.7% → 39.8%), T₂ˡ remains basically unchanged (0.222 → 0.223 ms), T₂ᵃᵛᵍ decreases (0.442 → 0.400 ms). Lysine acts as a crosslinker within or between ion aggregates via its amine and amino acid structure, increasing ion aggregate content while maintaining their rigidity, leading to decreased chain mobility.
Dimethylethanolamine (DMEA):
Aˡ increases (31.7% → 34.4%), T₂ˡ increases (0.222 → 0.257 ms), T₂ᵃᵛᵍ increases (0.442 → 0.504 ms). DMEA reacts with residual acid groups to form ammonium carboxylates, altering the structure of the original K⁺ carboxylate ion aggregates, reducing their rigidity, and enhancing chain mobility.
Dioctyl Phthalate (DOP):
Aˡ increases (31.7% → 37.2%), both T₂ⁱ and T₂ʰ increase (T₂ⁱ: 0.711 → 0.868 ms; T₂ʰ: 7.104 → 10.446 ms), T₂ᵃᵛᵍ slightly increases (0.442 → 0.471 ms). As a non-polar plasticizer preferring the PE phase, DOP reduces the polarity of the intermediate and high-mobility phases, inducing fusion of ion aggregates (intensified phase separation), thereby enhancing overall chain mobility.
Technical Value: TD‑NMR can intuitively distinguish whether a plasticizer “inserts into the ionic phase” or “enhances phase separation”, providing a rapid evaluation tool for screening plasticizers suitable for high-ion-content systems.
Summary: How Low-Field TD‑NMR Becomes a Powerful Tool to Address Pain Points in EMAA R&D and Quality Control
The population-weighted average T₂ value (T₂ᵃᵛᵍ) proposed in the study can directly serve as a quantitative indicator for EMAA chain mobility and processability, solving the challenge that traditional processability evaluations (like melt viscosity testing) are difficult to apply to high DoN (>60%) EMAA. Through changes in dynamic phase content (Aˡ, Aⁱ, Aʰ) and their respective T₂ values, the viscoelasticity, toughness, and other macroscopic properties of EMAA can be accurately predicted. This provides clear “microscopic parameter regulation” targets for polymer performance optimization, breaking away from the traditional “trial-and-error” R&D model.
The low-field TD‑NMR characterization protocol established in this study offers significant technical advantages: The total experimental time is only about 1 minute, avoiding the complex operations and long duration of high-field NMR. The three-component model fitting yields R² > 0.994, and the day-to-day variation for key parameters (T₂ᵃᵛᵍ) is < 2%, demonstrating excellent repeatability and stability, enabling adaptation from laboratory R&D to industrial testing. This technology system provides a “fast, precise, and low-cost” new solution for polymer dynamic characterization, promoting the application expansion of low-field NMR in the field of polymer materials.
References:
Chen, Z., Xu, Y., Zhang, Z., Yan, Q., Song, A., Qiu, X., Rau, N., & Drumright, R. (2025). Poly (ethylene-co-methacrylic acid) ionomer dynamic phase composition and polymer chain mobility characterization by time domain NMR. International Journal of Polymer Analysis and Characterization, 30 (7), 867-876. https://doi.org/10.1080/1023666X.2025.2462057
Phone: 400-060-3233
After-sales: 400-060-3233


Back to Top